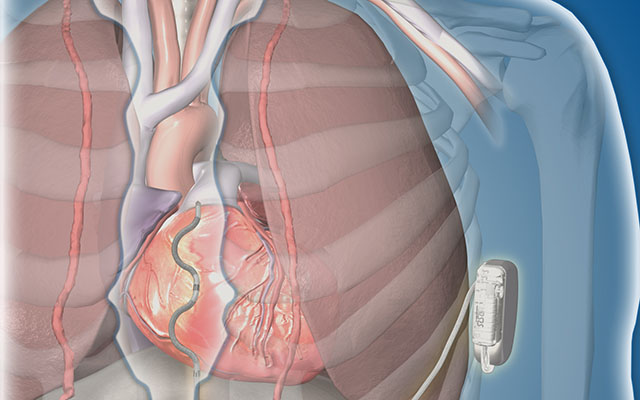
The investigational, implantable defibrillator system seeks to treat dangerously fast heart rhythms with a new technology
Cardiovascular physicians with UC San Diego Health have joined an international clinical trial utilizing a new Extravascular Implantable Cardioverter-Defibrillator (EV ICD) system to help treat sudden cardiac arrest. The first successful surgery was completed July 26, 2021.
Implantable cardioverter defibrillators (ICDs) have been used for more than 30 years to monitor and help regulate and pace an irregular heartbeat, which is often associated with greater risk of sudden cardiac arrest. Traditionally, these devices require implanting leads — thin wires that conduct electrical signals — inside the heart and blood vessels of patients. The new device being tested offers a different approach.
“With this new advancement in surgery, we’re no longer implanting these leads in a patient’s heart or vasculature, which we believe will result in better and safer care for patients at risk for sudden cardiac death,” said Ulrika Birgersdotter-Green, MD, director of Pacemaker and ICD Service at UC San Diego Health. “We’re excited to be a part of this clinical trial seeking to improve cardiovascular care for our patients.”
To prepare for surgery, the first patient to receive this device underwent a lead extraction procedure, a delicate procedure involving removal and replacement of the leads used in traditional ICDs. Pioneered at UC San Diego Health, physicians have now successfully performed nearly 1,000 such procedures.
This new, investigational, ICD device still requires leads, but rather than being implanted directly into the heart, the leads are placed outside the heart and veins. According to Birgersdotter-Green, this approach allows the device to offer more pacing, or heart stimulation, in the event of a sudden cardiac arrest.
“We’re moving away from placing leads in large blood vessels of a patient’s heart in a procedure that is innovative, completely outside the vasculature and also offers a small implant that is convenient for the patient,” said Birgersdotter-Green. “In turn, we’re providing a comprehensive care package that ultimately saves lives.”
The procedure is performed in the region’s first hybrid operating room — a state-of-the-art operating room that accommodates multiple types of treatment without moving the patient — located at the Sulpizio Cardiovascular Center at UC San Diego Health in La Jolla.
The clinical trial for the investigational device, developed by Medtronic, is a prospective, multicenter, single-arm, non-randomized, pre-market study evaluating the new technology in up to 400 patients at 60 hospitals in North America, Europe, the Middle East, Asia, Australia and New Zealand. UC San Diego Health is the second hospital system in California to take part in the trial.
This story was originally published by the University of California San Diego.