Chronic pain involves more than just hurting. People suffering from pain often experience sadness, depression, and lethargy. That’s one reason opioids can be so addictive—they not only dampen the pain but also make people feel euphoric.
What if it were possible to develop a painkiller that could curb the negative emotions associated with pain without causing euphoria? The new research is a step toward that goal.
The findings could lay the groundwork for developing new, less addictive approaches to pain treatment, researchers say.
“We’re in the midst of an opioid epidemic, and the euphoria associated with opioids is a major driver of opioid dependence,” says senior investigator Jose Moron-Concepcion, an associate professor of anesthesiology, of neuroscience, and of psychiatry at Washington University School of Medicine in St. Louis.
“By targeting the emotional aspects of pain, we hope to make pain less debilitating so that patients won’t crave the emotional high they get from opioids.”
THE BRAIN’S OPIOID RECEPTORS
Opioid painkillers, such as morphine, oxycodone, and fentanyl, target receptors on brain cells called mu opioid receptors. In contrast, the researchers studied kappa opioid receptors, which operate very differently.
Activating the kappa receptor makes people feel depressed, sad, and unmotivated. So Moron-Concepcion and his colleagues thought that by blocking those receptors, they also might dampen the negative emotions associated with pain.
For the study, researchers injected some of the rats in the paw with a substance that causes persistent inflammation. To measure the emotional effects of that pain, the researchers used a rewarding task in which the animals could work for sugar as a way to measure motivation. After being taught to push a lever to get sugar, most rats will keep pushing. In these experiments, the animals had to push the lever progressively more each time they wanted a pellet of sucrose.
“When the animals experienced pain, they were less motivated to work to obtain the reward,” says first author Nicolas Massaly, an instructor in anesthesiology. “It’s often the same for people in pain who don’t get as much pleasure from daily activities they usually enjoy.”
But when researchers treated the rats with inflamed paws with a compound to block kappa opioid receptors in their brains, the animals recovered the motivation to obtain the sugar, and pushed the lever as often as those who did not have inflamed paws.
RESTORING MOTIVATION
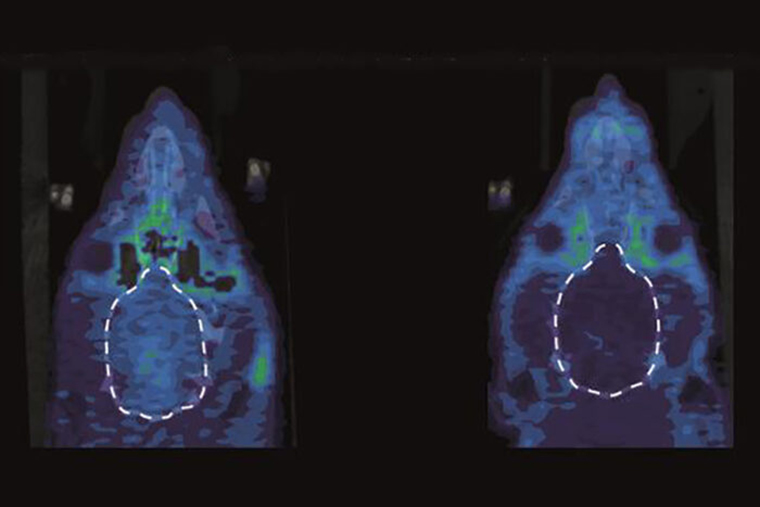
In addition, the researchers collaborated with Kooresh Shoghi, an associate professor of radiology, using small animal positron emission tomography (PET) imaging to assess the activity of kappa opioid receptors in the animals’ brains. They were able to demonstrate that when rats were in pain, their kappa opioid receptors were very active in a part of the brain—the nucleus accumbens—linked to emotion.
The researchers dampened this kappa opioid receptor activity by blocking the release of a natural stimulator of kappa opioid receptors called dynorphin—which the brain produces and is kind of like the inverse of the endorphins activities such as exercise release.
“By blocking dynorphin release, we were able to restore motivation in the animals despite the fact that we did not completely eliminate their pain,” Massaly says.
Moron-Concepcion, Massaly, and their colleagues acknowledge it’s a long journey from rodents to people. But they already have preliminary PET data from people, suggesting it may be possible to influence kappa opioid receptors and potentially prevent the sadness and lack of motivation that can accompany physical pain. They believe that by attacking pain’s emotional properties, without affecting the useful element of the pain response that can protect injuries from further damage, it may be possible to improve quality of life for pain patients without having to use any, or as many, habit-forming opioid pain killers.
Their findings are published in the journal Neuron.
The National Institute on Drug Abuse, the National Institute on Neurological Disorders and Stroke, and the National Institute on Alcohol Abuse and Alcoholism of the National Institutes of Health supported this work. Additional funding came from a NARSAD Independent Investigator Award, the Philippe Foundation, the Spanish Ministerio de Edonomia y Competitividad MINECO, and the Mallinckrodt Institute of Radiology.
By Jim Dryden
This story was originally posted by the Washington University in St. Louis.